![]() A few months ago, Chilean startup Copper3D introduced PLACTIVE, an antibacterial 3D printing filament designed for the production of medical devices such as prosthetics and braces. The company pointed to the worrying statistic that 40% of prosthetic uses suffer from some sort of skin condition, some of which can be severe or even life-threatening. PLACTIVE includes, among other materials, copper nanoparticles, which are active in the elimination of microorganisms, making devices 3D printed from the material effective in preventing infection.
A few months ago, Chilean startup Copper3D introduced PLACTIVE, an antibacterial 3D printing filament designed for the production of medical devices such as prosthetics and braces. The company pointed to the worrying statistic that 40% of prosthetic uses suffer from some sort of skin condition, some of which can be severe or even life-threatening. PLACTIVE includes, among other materials, copper nanoparticles, which are active in the elimination of microorganisms, making devices 3D printed from the material effective in preventing infection.
One organization that has taken an interest in PLACTIVE is NASA. Last year, the first medical devices were 3D printed in space, as astronauts at the International Space Station are moving toward the 3D printing of more and more necessities. Because of the porous nature of 3D printing materials, as well as the complex geometries of 3D printed devices, bacteria is always a concern, so NASA was intrigued by the notion of an antibacterial 3D printing filament.
The NASA Nebraska Space Grant is working with the University of Nebraska Omaha and Copper3D on a study of PLACTIVE.
“The use of this new antibacterial 3D printing technology has several potential applications,” said Daniel Martínez, Director of Innovation and Co-Founder of Copper3D. “Our main focus has been the development of antibacterial materials with applications in the field of Medicine and Rehabilitation, such as medical/surgical instruments, orthoses, prostheses, applications in the dental world, and healing of complex wounds. But now we want to go a step further and verify the potential of this technology in other types of challenges associated with the problems of the future of long-duration space missions.”
On long-duration space missions, prevention of infection is key in avoiding more serious health problems, and devices made from PLACTIVE could go a long way in keeping astronauts healthy after injury. The material has shown great promise on Earth so far.
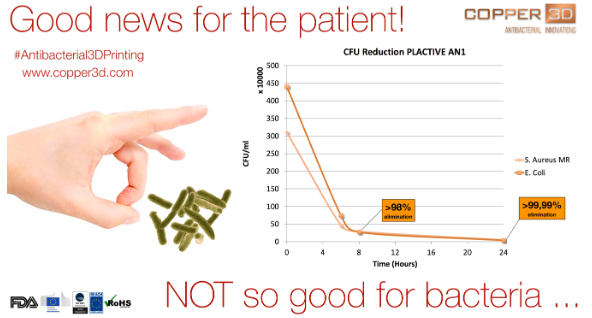
“This new material has already passed very exhaustive laboratory tests in Chile and the USA, with microbiological burden reduction studies that have demonstrated their ability to eliminate in a few hours +99.99% of dangerous bacterial strains such as Escherichia coli and Staphylococcus aureus, strains that are also very present in the hospital environment, causing millions of infections and deaths per year worldwide,” said Dr. Claudio Soto, Medical Director and Co-Founder of Copper 3D.
The scientific director of the study is Dr. Jorge Zuniga of the University of Nebraska Omaha, who has extensive experience in the design of 3D printed prosthetics.
“The objective of this research is threefold: i.-Describe the development of 3D printed prostheses with antibacterial filament, ii.-Verify the antibacterial properties of 3D printed prostheses and iii.-Develop a remote fitting methodology and determine patient satisfaction after using a 3D printed antibacterial finger prosthesis,” he said. “This study will also set the scientific basis for future studies on the impact of this new technology on countless new antibacterial applications that solve serious health and functional problems both on earth and in space.”
PLACTIVE has the potential to have a major impact on human health, and the current study is likely to lead to even more applications for the material.
“Our company started with a strong emphasis on innovation and our focus will continue to be linked to innovation and development of new materials and products that achieve a real impact on the quality of life of the people,” said Andrés Acuña, CEO and Co-Founder of Copper3D. “We are very excited about the results that may come out of this study with Dr. Jorge Zuniga, the University of Nebraska at Omaha and NASA. We think that this collaboration could be maintained over time to explore new technologies in the field of Additive Manufacturing that can save lives in complex contexts such as intra-hospital infections and environments as hostile as long duration space missions.”
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Unpeeled: New Arkema Material for HP, Saddle and Macro MEMS
A new Arkema material for MJF is said to reduce costs per part by up to 25% and have an 85% reusability ratio. HP 3D HR PA 12 S has been...
3D Printing News Briefs, January 20, 2024: FDM, LPBF, Underwater 3D Printer, Racing, & More
We’re starting off with a process certification in today’s 3D Printing News Briefs, and then moving on to research about solute trapping, laser powder bed fusion, and then moving on...
3D Printing Webinar and Event Roundup: December 3, 2023
We’ve got plenty of events and webinars coming up for you this week! Quickparts is having a Manufacturing Roadshow, America Makes is holding a Member Town Hall, Stratafest makes two...
Formnext 2023 Day Three: Slam Dunk
I’m high—high on trade show. I’ve met numerous new faces and reconnected with old friends, creating an absolutely wonderful atmosphere. The excitement is palpable over several emerging developments. The high...